Medical devices
From idea to patient — expert support at every stage
Are you looking for quality, regulatory or clinical support for your medical device? QbD Group can support you at any stage in the lifecycle of your medical device, from idea to patient. We are experts when it comes to ISO13485, clinical evaluation/investigation, MDR, design and development, technical files, auditing, qualification, validation, risk management, software, and more.
End-to-end lifecycle support
QbD Group provides expert guidance at every stage of your medical device's lifecycle, from concept to patient, ensuring compliance and market success.
Regulatory expertise you can trust
With in-depth knowledge of ISO 13485, MDR, and risk management, we help you navigate complex regulations to safely and effectively bring your device to market.
Comprehensive quality and clinical solutions
From establishing quality management systems to conducting clinical evaluations and investigations, QbD Group ensures your medical device meets the highest standards of safety and performance.
Post-market monitoring and maintenance
We support ongoing compliance by managing post-market monitoring and processes to maintain the safety and effectiveness of your medical device throughout its lifecycle.
Tailored solutions
Our solutions for Medical devices
Explore purpose-built solutions designed to address the specific regulatory, quality, and compliance challenges in your industry.
Cross-industry solutions
Your partner from idea to patient
Whether you're preparing for CE marking, navigating the MDD to MDR transition, or scaling post-market surveillance — our experts guide you through every stage of the medical device lifecycle.

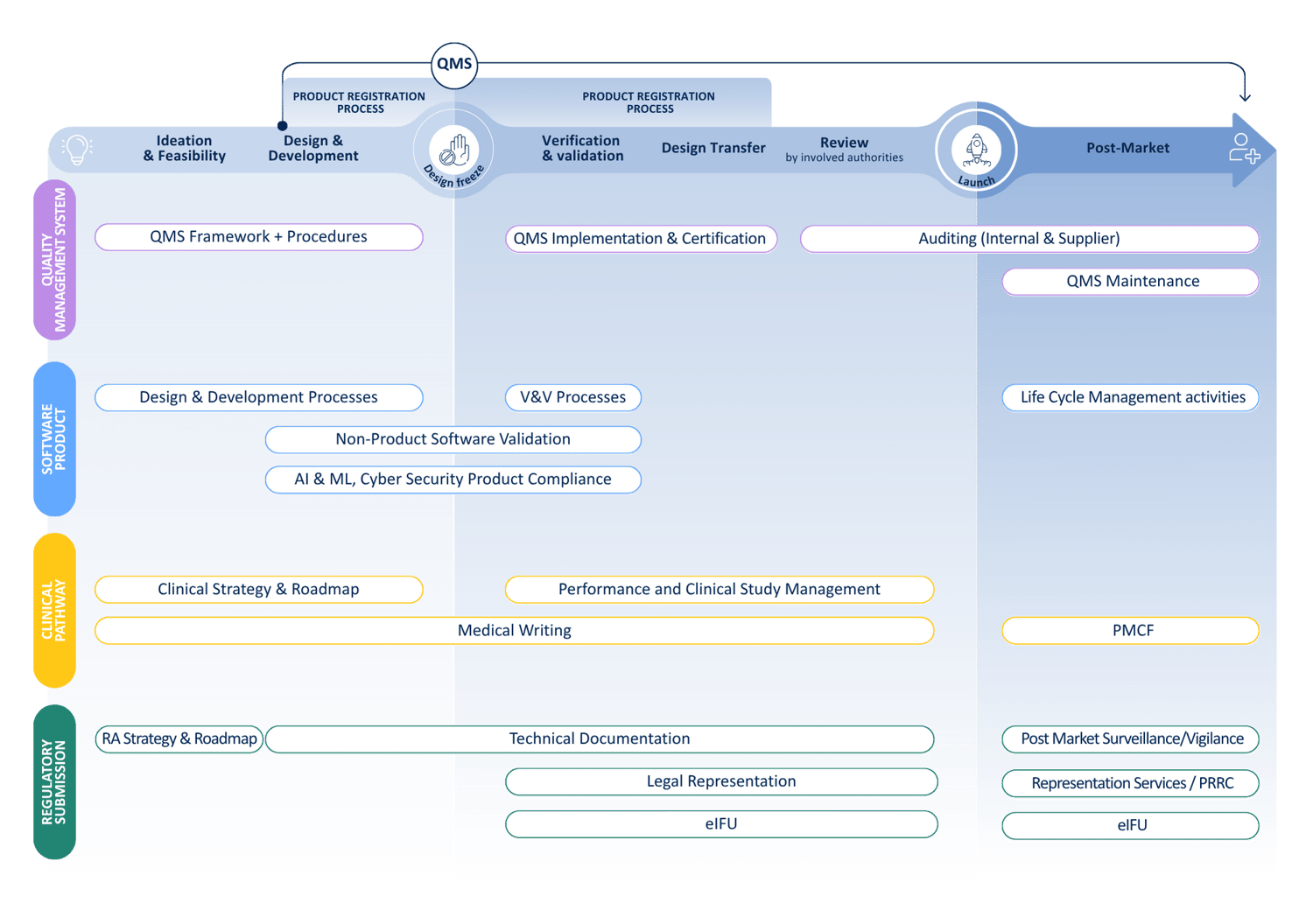
Lifecycle
Product lifecycle
Your partner at every stage — from concept and regulatory strategy through post-market compliance, we support the full Medical Device life cycle.
Our services
How we can help you4
Regulatory Affairs
- CE Marking, FDA clearance, and market entry strategies.
- Technical documentation and submission packages for MDR and global markets.
- PRRC services, in-country representation, legal representation, and post-market regulatory support.
Quality Assurance
- Implementation of FDA, EMA, FAHMP, and ISO standards (ISO13485, ISO14971, etc.)
- Internal and external audits by certified lead auditors.
- Lifecycle management for Medical Device Software and digital health solutions.
Clinical Services
- Strategy and protocol development for clinical evidence generation.
- Site selection, monitoring, and regulatory support.
- Comprehensive medical writing, incl. scientific validity reports and post-market clinical follow-up documentation.
Software Solutions & Services
- IEC 62304 software lifecycle compliance and design & development support.
- SaMD classification, AI Act readiness, and cybersecurity assessments.
- DevOps integration with QMS for streamlined software releases.
Services
Featured12

Why QbD Group
Your trusted partner
Within QbD Group, we combine in-depth regulatory, clinical, and quality expertise to help medical device manufacturers, distributors, and importers comply with the legislative requirements during the complete lifecycle of a medical device. From full-service clinical studies to technical documentation, our experts support you through every stage of the MDR certification process.
Trusted by Notified Bodies
our work quality is recognized by the key certification bodies in the industry.
Globally recognized MDR experts
with deep expertise across MDR, IVDR, and international regulations.
10+ years of experience
a proven track record in supporting medical device companies of all sizes.
On top of industry trends
we stay ahead of regulatory changes so you can focus on innovation.
FAQ
Frequently asked questions

Get in touch
Partner with QbD Group to ensure your medical device meets the highest standards of quality and compliance.