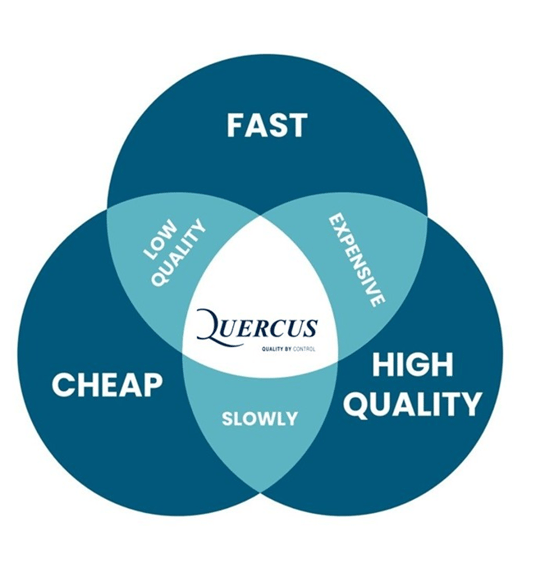
In every Good Manufacturing Practice (GMP) laboratory, three priorities continuously compete for attention: Quality, Cost, and Time.
Whether it’s a new product development project, a method validation campaign, or routine quality control, the so-called “ Triangle Dilemma” is always in play.
And while these three elements may seem like equal sides of a triangle, the reality is more complex: when one side is stretched, the others often feel the strain.
The Triangle Dilemma in GMP Laboratories
Quality: The Non-Negotiable Pillar
In the pharmaceutical industry, quality is not optional. Regulatory compliance, patient safety, and therapeutic efficacy rely on rigorous testing, validated methods, and clear documentation. Every result must be traceable, accurate, and defensible.
However, delivering this level of quality takes time—and resources. As compliance demands rise, so does the need for skilled staff, robust systems, and repeatable processes.
Cost: Constant Pressure to Do More with Less
No GMP lab operates in a vacuum. Budget constraints and market competition apply pressure to optimize costs. This might involve lean staffing, outsourcing routine tasks, or implementing automation. But cost savings must never jeopardize data integrity, traceability, or GMP compliance.
The real challenge? Knowing where to cut and where to invest.
Time: The Most Limited Resource
From fast-track clinical programs to tight batch release schedules, time is often the most critical constraint. Laboratories are expected to deliver results at speed—sometimes in hours, not days.
But speeding up without sacrificing quality takes more than urgency. It requires careful planning, sufficient lab capacity, and a well-coordinated, skilled team.
Balancing the Triangle: Practical Strategies
There’s no single solution to the triangle dilemma. Instead, successful labs rely on strategic decision-making and continuous improvement to strike the right balance. Here’s how:
1. Invest in Smart Technologies
Automation in sample prep, data processing, and reporting can improve speed and consistency while reducing human error. Tools like LIMS and chromatography data systems help standardize workflows and improve data integrity.
2. Apply Risk-Based Thinking
Not every test or decision carries the same weight. Using ICH Q9 principles, labs can prioritize resources based on risk and criticality, focusing effort where it has the biggest impact.
3. Streamline Workflows
Lean lab practices, digital tools, and process mapping help eliminate non-value-added steps. The result: shorter lead times, better transparency, and fewer bottlenecks.
4. Strengthen Training and Flexibility
Cross-functional training equips staff to switch roles during peak periods. A resilient team is key to managing high volumes while maintaining compliance.
5. Outsource Strategically
Partnering with qualified Contract Research Organizations (CROs) for non-core or high-volume activities can ease internal pressure—provided partners uphold GMP standards and integrate well with internal QA systems.
Conclusion: A Strategic Balancing Act
The triangle dilemma won’t disappear—but it can be managed. The key is alignment: ensuring that quality expectations, budget constraints, and timeline pressures are transparent and accounted for from the outset.
Ultimately, it’s not about finding the perfect balance. It’s about making informed trade-offs that protect product quality, ensure compliance, and support business goals.
Quercus, part of the QbD Group, helps pharmaceutical companies implement smart lab strategies that combine compliance, efficiency, and agility.
Don't hesitate to get in touch.

Need reliable lab testing and validation?
About the Author
MP, QP · Division Head Lab Services
Yves leads the Lab Services division at QbD Group, bringing deep expertise in GMP laboratory operations, analytical method validation, and quality control for pharmaceutical manufacturing.
Subscribe to the latest updates in life science
Expert perspectives delivered to your inbox — pick your interests.
No spam, ever. Unsubscribe anytime.



